The Kinetic Supply Chain: Understanding the Radiopharmaceutical Reality
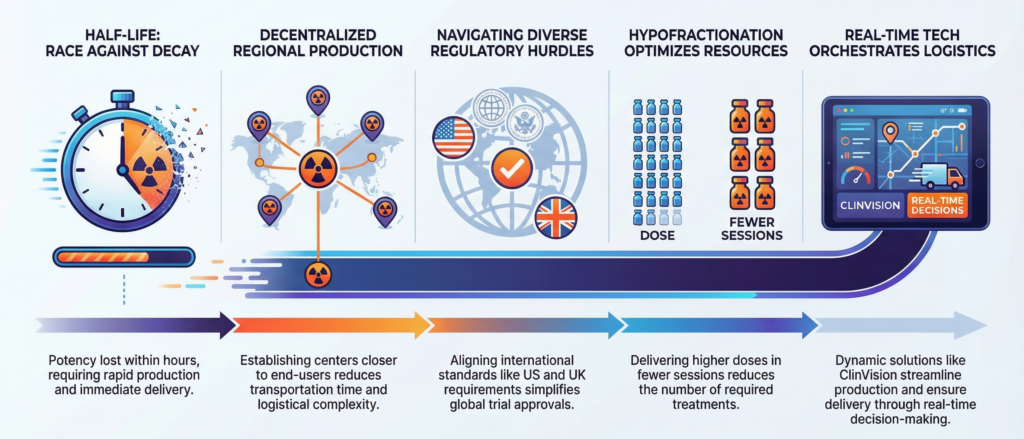
In this sector, “decay” is more than a loss of potency; it is the fundamental metamorphosis of the product itself. Because these isotopes have half-lives ranging from a few hours to a few days, the supply chain faces constant pressure for accelerated timelines. There is no “standing inventory.” Every dose is effectively manufactured for a specific patient at a specific moment.
The Logistics of Decay

Category | Traditional Pharmaceuticals | Radiopharmaceuticals |
Shelf-life | Long (months to years); chemically stable. | Extremely short (hours to days); physical decay. |
Delivery Urgency | Standardized; supports warehousing and stockpiling. | Immediate; requires just-in-time delivery post-production. |
Handling Requirements | Standardized chemical safety protocols. | Specialized shielding and strict radiation safety mandates. |
Planning Horizon | Weeks or months of lead time. | Real-time or hour-by-hour synchronization. |
To mitigate the risks of this short shelf-life, the industry utilizes two integrated strategies. Decentralized Production involves establishing a network of regional manufacturing hubs situated in close proximity to end-users. This physical infrastructure serves as the essential foundation that enables a Real-time Reactive Command structure. By reducing the geographical distance between the cyclotron or generator and the clinic, logistics providers can operate a highly synchronized supply chain that reacts instantly to manufacturing outputs, ensuring the medicine reaches the patient before its therapeutic window expires.
While the physical instability of the isotope dictates the velocity of the supply chain, the movement of these materials is ultimately governed by a complex, multi-layered regulatory environment.
The Regulatory Maze: Comparing USA and UK Frameworks
Navigating clinical trial approvals for radiopharmaceuticals requires satisfying both health-related and nuclear-safety-related authorities. This dual-oversight creates a sequential and often rigorous path to site activation.
Comparative Approval Checklist The specific requirements for initiating clinical trials demonstrate significant jurisdictional variance:
- United States (USA):
- Radiation Safety Committee (RSC) Approval: This is a mandatory first step. RSC approval regarding radiation dose and safety protocols must be secured before the study can be submitted to the Institutional Review Board.
- Institutional Review Board (IRB) Submission: Once the RSC has cleared the safety profile, the IRB performs the standard ethical and clinical review.
- United Kingdom (UK):
- ARSAC Authorization: Researchers must obtain authorization from the Administration of Radioactive Substances Advisory Committee.
- Ethics Review: In the UK, the ARSAC review and ethics review typically occur as mutual, parallel efforts to ensure patient safety and scientific validity.
The Strategic Value of Regulatory Harmonization In an era of globalized clinical development, “regulatory harmonization” has become a strategic necessity rather than an abstract ideal. Mutual efforts to align international standards—such as cross-border collaboration on radiation safety benchmarks—are critical for simplifying the approval process. Harmonization reduces the bureaucratic friction that currently delays global trials, facilitating more equitable patient access to emerging theranostic innovations.
As regulators strive to streamline these legal frameworks, clinical practitioners are simultaneously evolving their treatment models to maximize facility throughput and patient outcomes.
Building the Future: Workforce, Technology, and Scalability
The primary obstacles to scaling nuclear medicine remain the human and technical bottlenecks of staff shortages and zero-tolerance quality compliance. Because radiopharmaceuticals are subject to an accelerated timeline, there is no margin for error; a logistical delay is not just a setback, but a total loss of product.
To address these vulnerabilities, specialized technology such as S-CLINICA’s ClinVision (TrackMed module) is being integrated into the manufacturing ecosystem. These solutions provide three core capabilities:
- Advanced Planning: Allows sponsors to map the entire manufacturing ecosystem and supply chain in advance.
- Primary Benefit: Mitigates technical bottlenecks and optimizes resource allocation to reduce operational costs.
- Dynamic Communication: Establishes a real-time data bridge between the manufacturer, the courier, and the clinic.
- Primary Benefit: Supports quality compliance by ensuring the production schedule is perfectly synchronized with patient arrival, eliminating the risk of isotopic expiration.
- Real-time Decision Making: Provides instantaneous data visibility throughout the trial lifecycle.
- Primary Benefit: Empowers managers to intervene immediately during logistical disruptions, ensuring successful delivery despite the “short shelf-life” pressure.
- Primary Benefit: Empowers managers to intervene immediately during logistical disruptions, ensuring successful delivery despite the “short shelf-life” pressure.
Furthermore, technology must be paired with human expertise. S-CLINICA’s expert support and credentialing initiatives are vital for building a robust global team. By providing standardized training and certification, these initiatives address the global workforce shortage and ensure that personnel across the supply chain are equipped to handle these specialized materials safely and efficiently.
Conclusion: Towards a Resilient Ecosystem
The transition of nuclear medicine from a niche field to a cornerstone of modern oncology requires a move away from fragmented, ad-hoc logistics. The industry must commit to the Three Pillars of a Future-Ready Ecosystem:
- Scalable Infrastructure:Expanding decentralized manufacturing hubs to support regional growth.
- Streamlined Regulatory Pathways:Reducing global friction through strategic cross-border collaboration and mutual standards.
- A Skilled Workforce:Prioritizing investment in credentialing and technical training to support a growing clinical demand.
By integrating these disparate elements—advanced logistics, harmonized law, and real-time technology—the industry can transform current limitations into a resilient, future-ready ecosystem. Strategic investment in these areas is the only way to ensure that the life-saving potential of nuclear medicine is fully realized on a global scale.